Buckyballs and Nanotubes: Difference between revisions
m (Text replace - "[[Category:Computers and information processing" to "[[Category:Computing and electronics") |
m (Text replace - "[[Category:Engineered_materials_&_dielectrics" to "[[Category:Materials") |
||
| Line 17: | Line 17: | ||
[[Category:Computing and electronics]] | [[Category:Computing and electronics]] | ||
[[Category:Integrated_circuits]] | [[Category:Integrated_circuits]] | ||
[[Category: | [[Category:Materials]] | ||
[[Category:Elements]] | [[Category:Elements]] | ||
[[Category:Carbon]] | [[Category:Carbon]] | ||
Revision as of 15:20, 25 July 2014
Buckyballs and Nanotubes
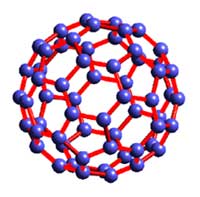
“Buckyball” is the common name for a molecule called Buckminsterfullerene, which is made of 60 carbon atoms formed in the shape of a hollow ball. British scientist Harry Kroto discovered it in 1985. The arrangement of the atoms resembled the shape of the geodesic domes invented by architect Buckminster Fuller, hence the name. Along with American researchers Richard Smalley and Robert Curl, Kroto was able to create buckyballs in the laboratory. For this work, the three were awarded a Nobel Prize in Chemistry in 1996. These researchers found that buckyballs had many interesting properties. They were very hard to break apart, even at temperatures of nearly 1000 °C, and would bounce back if squeezed or slammed against a solid object.
Buckyballs have not yet found many practical applications, but research by Sumio Iijima in 1991 led to the discovery of buckytubes, now more commonly called nanotubes. These are tubes of carbon atoms, about a nanometer in diameter (one billionth of a meter), which conduct electricity and which are stronger than steel. The first nanotubes consisted of tubes-within-tubes, but later chemists found ways to form tubes with just one layer and a hollow space inside. Nanotube walls have the same “soccer ball” structure as the buckyball, but they are rolled up into long tubes. The tubes can be up to a millimeter long and contain over 1,000,000 atoms. This is all very interesting, but just what are buckyballs and nanotubes good for?
Researchers have spent the last 15 years finding uses for buckyballs and nanotubes. Today, nanotubes are used as the stylus in atomic force microscopes (AFMs). Nanotubes are also being used as filters to separate extremely small objects from liquids or the air. Because they have hollow cores, small molecules can pass through them but larger objects, such as bacteria and viruses, cannot.
Both buckyballs and nanotubes conduct electricity well, but later research also revealed that under certain conditions, nanotubes will conduct in only one direction, like a semiconductor diode. After several years of work, it was possible to demonstrate in the laboratory nano-scale electronic circuits with wires and switches made from nanotubes. These nano-components can be arranged into individual memory “cells,” that simulate the circuit used to store one bit of information on a semiconductor integrated circuit. In the next ten to twenty years, engineers hope to be able to make more complex integrated circuits, such as large-scale memories or microprocessors.
Categories: Engineered materials & dielectrics | Components, circuits, devices & systems | Nanotechnology | Nanostructures | Elements | Carbon | Integrated circuits | News